Health is a prime objective in today’s world, and everything related to it is strictly tested to ensure its use is safe and effective. Similarly, medical devices also pass through strict standards set by the respective healthcare authorities before any facility uses them. In the UAE, these standards are set by the federal authority, MOHAP, supported by the local authorities, including DHA, DOH, and SHA, through the Drug Control Department.
Medical devices, after complying with those standards, can be registered by UAE-based manufacturers or authorized local representatives. The standards mainly include English documentation, mandatory vigilance systems, adverse event reporting, and recall procedures to ensure compliance and patient safety. However, before device registration, the facility or company must obtain an MOHAP license to import, use, or market products.
The registration process depends on device classification, which is mainly divided into 4 categories, including Class I, Class IIa, Class IIb, and Class III. Once a device is registered, you can legally import, use, and sell it. The process includes appointing a Local Authorized Representative, creating an MOHAP account, submitting device classification and documentation, undergoing regulatory review and evaluation, and receiving the approval and registration certificate.
The registration normally takes from 15 to 45 working days, while the cost depends on the product category and whether the company already has a trade license or not. The time also depends on the authority regulation and document preparation. Missing or incorrect data will delay the approval and increase the estimated cost.
However, you can avoid these by keeping clear device classification, complete technical files, and compliance with the MOHAP device registration regulations. Healthcare license consultancies in the UAE, like Felix Happich and DataFlow, can help you with the process by ensuring clear device classification, preparing documents correctly, and following rules and regulations through local knowledge.
What is Medical Device Registration in the UAE?
Medical device registration in the UAE is the mandatory authorization process from healthcare authorities (MOHAP, DHA, and DOH), through which medical devices are approved before they can be legally imported, marketed, or used within the UAE, as per ISO standards.
Medical device registration helps ensure the effectiveness and safety of the device, keeping patients safe and providing them with quality care. The process applies to a wide range of products, including diagnostic equipment, surgical instruments, implants, and digital health devices.
Why is Medical Device Registration Important in the UAE?
Medical device registration is important in the UAE to ensure patient safety, quality, and efficacy by meeting the strict regulatory standards set by the MOHAP, thereby fulfilling international healthcare standards. It is also crucial for preventing unsafe products from harming users and for allowing authorities to track products, conduct audits, and initiate recalls.
Key Reasons for Medical Device Registration:
- Ensure patient safety
- Maintain compliance with UAE healthcare laws
- Prevent the unfair use of medical devices
- Maintaining legal import and export
- Building among facilities and patients
Which Authority Regulates Medical Device Registration in the UAE?
Medical device registration in the UAE is primarily regulated by the Ministry of Health and Prevention (MOHAP) through the Drug Control Department as a federal authority, while the DHA and DOH also set standards to oversee the use and compliance of these devices in line with healthcare standards in individual emirates. The authority checks the device’s safety and quality by verifying the documents and certifications, as well as proper labeling and packaging, before it is imported or exported.
How Are Medical Device Registrations Classified in the UAE?
Medical devices are classified based on the Ministry of Health and Prevention (MOHAP) and the Emirates Drug Establishment (EDE), with support from the Dubai Health Authority, based on their intended use, level of contact with the body, duration of use, implantability, and their impact on the patient’s body.
Main Classification System (Class I → IV)
In this classification system, medical devices are categorized by risk level. These include.
- Class I – Low risk
- Class IIa – Low to medium risk
- Class IIb – Medium to high risk
- Class III – High risk
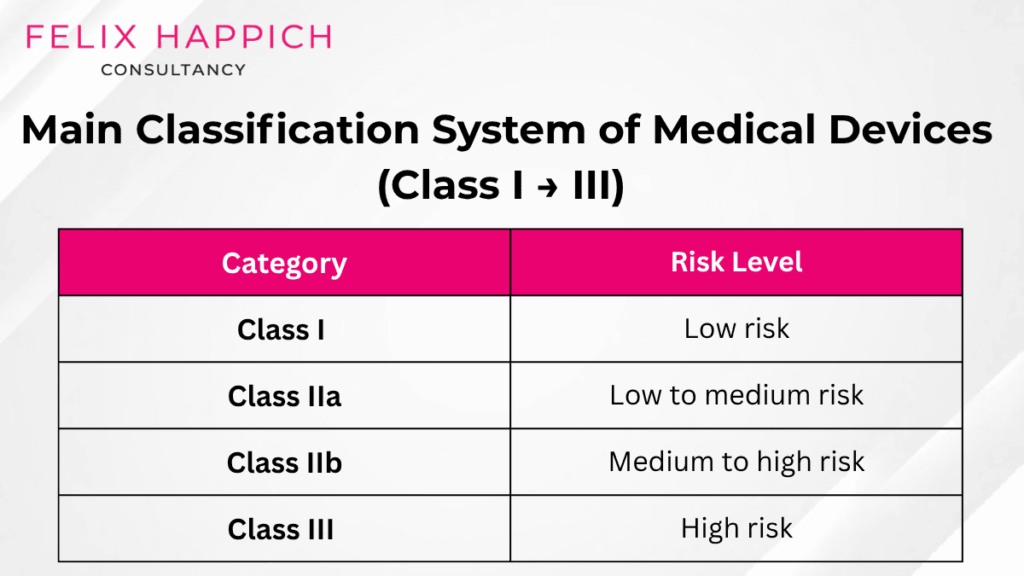
The table below shows a detailed example of each category.
| Class | Description | Examples |
|---|---|---|
| Class I | Non-invasive and has minimal impact | Bandages, thermometers, stethoscopes |
| Class IIa | limited invasion and has a moderate impact | Blood pressure monitors, ultrasound machines, and infusion pumps |
| Class IIb | Invasive and provides critical function support | Dialysis machines, orthopedic implants, ventilators |
| Class III | Life-supporting devices | Pacemakers, heart valves, and implantable defibrillators |
IVD Classification System
The In Vitro Diagnostic (IVD) classification system groups medical diagnostic devices based on their level of risk to patients and public health. Lower-risk devices need simpler checks, while higher-risk devices require strict evaluation. Below is the breakdown of the IVD classifications system.
| IVD Class | Risk Level | Examples |
|---|---|---|
| Class A | Low risk | Sample containers, buffer solutions |
| Class B | Low to moderate risk | Pregnancy tests, urine test strips |
| Class C | Moderate to high risk | Blood glucose tests, HIV screening tests |
| Class D | High risk | Blood screening for HIV/Hepatitis, donor tests |
What are the UAE medical device registration requirements?
The requirements for medical device registration in the UAE mainly include having reference country approvals, appointing an LAR, proper device classification (Class I → IV), and submitting technical documentation (ISO 13485 and Free Sale Certificate).

Other key requirements are provided below.
- A device company should have reference country approvals
- A valid MOHAP license for the facility to use the device
- Appoint a Local Authorized Representative (LAR) in the UAE
- Device must be approved in a recognized manufacturing country (e.g., US, EU, UK)
- Device quality should comply with a Quality Management System (QMS), such as ISO certifications (e.g., ISO 13485).
- Submit complete technical documentation and instructions in English and Arabic
- Provide proof of safety and performance
- Provide documentation of the manufacturing site
What Documents Are Required?
The main documents required for device registration include:
- Device description document and Labeling card
- A valid and active certificate of free sale (CFS)
- Instructions for Use (IFU) in English and Arabic
- ISO certification (e.g., ISO 13485)
- Classification report card with proper risk details
- Authorization letter for the Local Authorized Representative
What is the process for medical device registration in the UAE?
The process for medical device registration in the UAE includes registering the company and the device manufacturer with the Ministry of Health and Prevention. After manufacturer registration, it appoints a Local Authorized Representative and issues an authorization letter that allows them to legally handle the process on behalf of the manufacturer.
The applicant then submits the device classification and technical documents online. Once all required documents are submitted to MOHAP, it reviews the application and, upon approval, issues a registration certificate allowing you to legally use the device within the emirate. Below is the UAE medical device registration process in five clear steps:

- Appoint an LAR
- Create an MOHAP account
- Submit required documentation
- Undergo MOHAP review
- Receive the device registration certificate
Appoint an LAR
This is the first step, where the manufacturer appoints a representative who has a licensed local company. The manufacturer issues a signed authorization letter, allowing the appointed Local Authorized Representative to legally submit applications and communicate with the Ministry of Health and Prevention on its behalf.
Create an MOHAP account
This is the second step, and it is for the Local Authorized Representative, which involves creating an account on the Ministry of Health and Prevention portal using UAE PASS. This account is used to proceed with the online device registration process and to track the application status. It also helps in communication with the regulatory authority during the medical device registration process.
Submit required documentation
The third step is submitting the required device documentation, including the device classification based on risk level, device description, intended use, labeling, and quality documents like ISO 13485 to support safety and performance.
Undergo MOHAP review
The fourth step of the registration process is that the Ministry of Health and Prevention reviews the submitted application and checks the technical documents to ensure the device is safe and complies with global device quality standards.
Receive the device registration certificate
This is the final step after the Ministry of Health and Prevention reviews and considers the device suitable for registration, issuing a medical device registration certificate. This certificate is typically valid for five years and allows the device to be legally marketed, distributed, or used within the UAE.
How Long Does the UAE medical device registration Process Take?
The UAE medical device registration process usually takes between 3 and 6 months, depending on the device classification, document preparation, and the appointed Local Authorized Representative. For example, Class I devices may take approximately 2–4 months, while Class II devices may take around 4–6 months. The typical registration times for all the main categories of medical devices in the UAE are outlined below.
How Much Does Medical Device Registration Cost?
Medical device registration costs vary depending on device classification, licensing fees if not yet licensed, and Local Authorized Representative (LAR) service fees. On average, it costs around AED 5,500 if the manufacturer has an MOHAP license. Otherwise, the total cost with the license can reach AED 15,000–16,000. Below are the approximate cost points.
| Process | Fee (AED approx.) |
|---|---|
| Product Classification | 500 AED |
| Manufacturer Registration License | 10,500 AED |
| Product Registration | 5,000 AED |
What Are the Common Challenges in Registering a Medical Device in the UAE?
Below are the four common challenges in registering a Medical Device in the UAE include.
- Incomplete or incorrect documentation
- Misclassification of devices
- Difficulty appointing a qualified LAR
- Lack of prior approval in reference markets
How Can Felix Happich Help You with Device Registration?
Just like the UAE healthcare licensing services, Felix Happich also provides medical device registration services, managing the full registration process and improving approval efficiency. We organize complete documentation, help you prepare accurate and complete documents, confirm device classification early, and maintain ISO quality compliance.
FAQs About Medical Device Registration in the UAE
Who is responsible for medical device registration in the UAE?
The Ministry of Health and Prevention (MOHAP) is the primary authority responsible for medical device registration across the UAE.
Do foreign manufacturers need to register medical devices in the UAE?
Yes, foreign manufacturers need to register medical devices in the UAE before selling them.
Can distributors register medical devices on behalf of manufacturers?
Yes, distributors can register devices if they are officially appointed as the Local Authorized Representative.
Do all clinics need to register medical devices before using them?
Clinics do not register devices themselves, but they must ensure that all devices they use are properly registered and approved.
Is a UAE company required to register medical devices?
Yes, a UAE-based entity (LAR or distributor) is required to submit and manage the registration process.
What are the requirements for a Local Authorized Representative (LAR)?
The requirements for a Local Authorized Representative (LAR)
- Must have MOHAP license
- Must have regulatory knowledge and experience
- Must be authorized by the manufacturer
Is local testing required in the UAE?
In most cases, local testing is not required if the device is already approved in recognized international markets. However, authorities may request additional verification if needed.
How Long Is the Medical Device Registration Valid in the UAE?
Medical device registration in the is generally valid for 5 years, after which it must be renewed.





